Health Plans
Focused on Lower Costs for Payers and Patients and Better Outcomes
Navis Clinical Laboratories® works collaboratively with organizations to provide programs and reporting to optimize the use of lab services for provider networks and health plan members. Our goal is to support medication management services to reduce out-of-network leakage and drive appropriate testing utilization.
Exceptional Compliance Program
Navis’ robust compliance program ensures all business practices are consistent with applicable legal and regulatory mandates and meet all elements of the OIG Model Compliance Program for Clinical Laboratories.
Based on quarterly reviews led by our compliance team, Navis team members conduct in-person meetings with various clinicians to review aggregate drug testing results, including discussions on:
- Drug testing frequency, review of the practice’s positivity rates, medical necessity to support requirements, medical record documentation
- Current health plan policies and how they relate to existing test ordering patterns
Lower Costs for Payers
Navis’ program helps better monitor patient test frequency. From 2016-2018, Tier 1 confirmations increased from 56.5% to 74.1%, while Tier 4 confirmations decreased from 17.5% to 1.4%.*
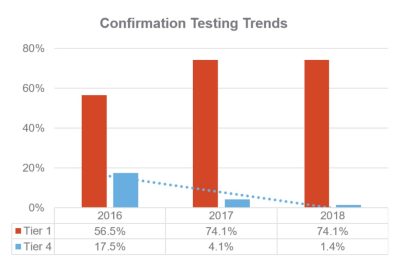
*n=15,351 tested claims from single payer, 2016-2018
Full-Service Testing Capabilities
Navis’ extensive testing capabilities support the ability to choose the right test at the right time.
Insurance Networks and Payment
Our laboratories are in-network with insurance providers nationwide and partner with new payers regularly to eliminate unnecessary healthcare costs. We also maintain electronic data interfaces (EDIs) with nearly all of our payers to avoid processing errors.
Beyond Toxicology Testing
Navis’ solutions go beyond improving patient safety and outcomes to help your organization reduce fraud, abuse and waste, manage high-cost cases, and utilize healthcare dollars more effectively.
Certifications
We maintain certifications from the Clinical Laboratories Improvement Amendments (CLIA) and the College of American Pathologists (CAP) to not just meet but exceed industry quality standards.
