Six Facts About Testing for Ethyl Glucuronide (EtG)
Like any other toxin, ethanol is excreted from the body in a variety of ways. These include:
- Direct excretion of ethanol (5–10%) in urine, sweat and breath
- Metabolic excretion by conversion to acetaldehyde/acetic acid (> 90%)
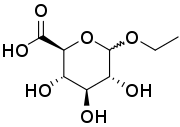
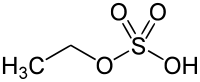
- Metabolic excretion by conversion to ethyl glucuronide and ethyl sulfate (< 0.1%), both of which are readily eliminated through urination
While most of these excretory products are detectable in urine for very short periods of time (less than 24 hours), ethyl glucuronide (EtG) and ethyl sulfate (EtS) have longer half-lives and may be detectable in urine for up to 80 hours post-consumption at industry-standard cutoffs, depending on the dose taken prior to specimen collection.
Many common products purchased at grocery stores or pharmacies contain ethanol, which can cause a positive result in a drug test for EtG.
These products include over-the-counter, alcohol-based cold and flu medications (e.g., NyQuil), food products (e.g., sauerkraut, balsamic vinegar, vanilla extract) and personal products such as mouthwashes. Additionally, certain popular drinks sold in stores (e.g., kombucha) may contain significant amounts of alcohol. It is also important to know that supposed non-alcoholic beverages may contain up to 1% ethanol by volume.
A diabetic with a specific urinary tract infection represents perhaps the most common example of in-vitro formation of ethyl glucuronide in the body. If the patient’s diabetes is uncontrolled, excess glucose in the blood is excreted into the urine and can be fermented into alcohol by microbial organisms. If E. coli, a predominant strain of bacteria often causative of urinary tract infections, is present in the urine, the fermented ethanol may be converted to ethyl glucuronide and be present in measurable amounts. This process occurs in diabetics only.
EtG is subject to bacterial degradation if the urine specimen is stored at room temperature for too long after collection. When degradation occurs, the amount of EtG in the urine decreases over time. In these cases, a sample that may have initially been positive may have significantly lower EtG levels if it is reanalyzed at a later date. Published scientific literature has shown EtS to be more stable than EtG at room temperature. Due to the inherent instability of EtG in urine specimens at room temperature, Cordant Health Solutions recommends that specimens intended for EtG testing be refrigerated if they cannot be shipped to the proper laboratory within three days. In addition, EtS is measured and reported on any specimen that is positive for EtG.
While higher amounts of EtG might indicate greater alcohol consumption, the exact EtG number is influenced by several factors, including how recently alcohol was consumed. The presence of EtG in urine indicates only that the individual was exposed to ethanol at some point in the recent past prior to testing, typically within the preceding 80 hours. No scientific correlation has been established between urine EtG levels and either blood or breath alcohol levels. Additionally, no states or jurisdictions have officially defined the legal limits for urinary EtG levels.
EtG testing has been presented as evidence in court proceedings and hearings. EtG testing has been subjected to, and passed, scientific admissibility standards (a process known as a Frye hearing) in several court cases.
The information provided is for educational purposes only and is not intended to be used as medical or legal advice. Detection times are approximate and may be influenced by a number of factors, including BMI, duration of drug use, dosage and interindividual metabolic differences. For specific drug testing result interpretation questions, please contact us.